Lenvatinib Impurity 10 | CAS 2817417-36-8 - Request Quote
Lenvatinib Impurity 10
| SZ CAT No: | SZ-L038012 |
| CAS No | 2817417-36-8 |
| Mol.F. | C19H16ClN3O5 |
| Mol.Wt. | 401.8 |
| Inv. Status | In Stock |
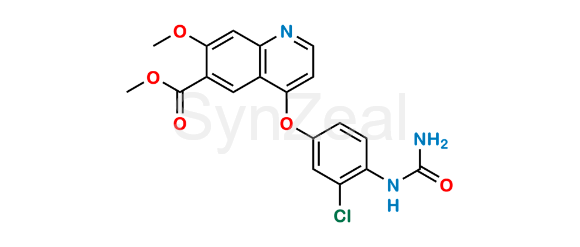
Chemical Name :
Shipping Temperature :
HSN Code :
Country of Origin :
Smiles :
CHNS Ratio (%) :
Usage Note:
Lenvatinib Impurity 10 is chemically Methyl 4-(3-chloro-4-ureidophenoxy)-7-methoxyquinoline-6-carboxylate. Lenvatinib Impurity 10 is supplied with detailed characterization data compliant with regulatory guideline. Lenvatinib Impurity 10 can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Lenvatinib.
The product can be used as reference standards and further traceability against pharmacopeial standards (USP or EP) can be provided based on feasibility. SynZeal products are for analytical purpose only and not for human use.
Applications & Regulatory Use Cases
Buy 2817417-36-8
Purchase 2817417-36-8
Order 2817417-36-8
Enquire 2817417-36-8
price of 2817417-36-8
2817417-36-8 Supplier
2817417-36-8 Manufacturer
2817417-36-8 Exporter
buy high quality Lenvatinib Impurity 10
Purchase Lenvatinib Impurity 10
Lenvatinib Impurity 10 suppliers
Lenvatinib Impurity 10 manufacturers
Lenvatinib Impurity 10 price
Order Lenvatinib Impurity 10
Enquire Lenvatinib Impurity 10
Lenvatinib Impurity 10 cost
Lenvatinib Impurity 10 Supplier
Lenvatinib Impurity 10 Distributor
Lenvatinib Impurity 10 for Method Validation
Lenvatinib Reference Standard
Lenvatinib Impurity 10 for ANDA Filing
Lenvatinib Impurity 10 for Forced Degradation Studies
Lenvatinib Impurity 10 Identification Standards
Lenvatinib Impurity 10 for DMF Filing
Related Products
Disclaimer
SynZeal product information given on this website is as per the existing understanding while publishing the details on website. The customer is responsible for assessing the accuracy of the information at the time of actual purchase.
SynZeal will update these details as per new developments or findings in product specifications without further notice.




