N-Nitroso Cilazapril EP Impurity B - 请求报价
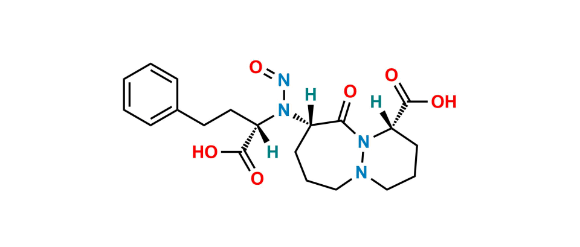
化学名称 :
运输温度 :
HSN编码 :
原产国 :
微笑 :
CHNS 比例 (%) :
使用说明:
N-Nitroso Cilazapril EP Impurity B is chemically (1S,9S)-9-(((S)-1-Carboxy-3-phenylpropyl)(nitroso)amino)-10-oxooctahydro-6H-pyridazino[1,2-a][1,2]diazepine-1-carboxylic acid. N-Nitroso Cilazapril EP Impurity B is supplied with detailed characterization data compliant with regulatory guideline. N-Nitroso Cilazapril EP Impurity B can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Cilazapril.
The product can be used as reference standards and further traceability against pharmacopeial standards (USP or EP) can be provided based on feasibility. SynZeal products are for analytical purpose only and not for human use.
应用与法规使用场景
buy high quality N-Nitroso Cilazapril EP Impurity B
采购 N-Nitroso Cilazapril EP Impurity B
N-Nitroso Cilazapril EP Impurity B 供应商
N-Nitroso Cilazapril EP Impurity B 制造商
N-Nitroso Cilazapril EP Impurity B 价格
订购 N-Nitroso Cilazapril EP Impurity B
询问 N-Nitroso Cilazapril EP Impurity B
N-Nitroso Cilazapril EP Impurity B 成本
N-Nitroso Cilazapril EP Impurity B 供应商
N-Nitroso Cilazapril EP Impurity B 分销商
N-Nitroso Cilazapril EP Impurity B 用于方法验证
Cilazapril 对照标准
N-Nitroso Cilazapril EP Impurity B 用于ANDA申报
N-Nitroso Cilazapril EP Impurity B 用于强制降解研究
N-Nitroso Cilazapril EP Impurity B 鉴别标准
N-Nitroso Cilazapril EP Impurity B 用于DMF申报
相关产品
免责声明
SynZeal 在本网站上提供的产品信息是根据发布时的现有理解而提供的。客户在实际购买时有责任评估信息的准确性。
SynZeal 将根据产品规格的新发展或新发现更新这些详细信息,恕不另行通知。




