Celiprolol Hydrochloride EP Impurity F | CAS 79881-89-3 - Solicitar cotización
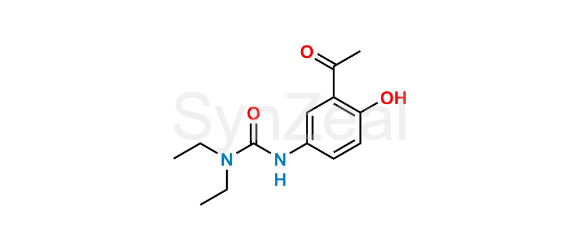
Celiprolol Hydrochloride EP Impurity F
| N° de SZ CAT: | SZ-C131007 |
| Número CAS | 79881-89-3 |
| Mol.F. | C13H18N2O3 |
| Peso Molecular | 250.3 |
| Status de Fatura | In Stock |
Nome Químico :
Temperatura de envío :
Código HSN :
País de origen :
Sorrisos :
Proporción CHNS (%) :
Nota de Uso:
Celiprolol Hydrochloride EP Impurity F is chemically N-(3-Acetyl-4-hydroxyphenyl)-N′,N′-diethylurea (as per EP). Celiprolol Hydrochloride EP Impurity F is supplied with detailed characterization data compliant with regulatory guideline. Celiprolol Hydrochloride EP Impurity F can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Celiprolol Hydrochloride.
The product can be used as reference standards and further traceability against pharmacopeial standards (USP or EP) can be provided based on feasibility. SynZeal products are for analytical purpose only and not for human use.
Aplicaciones y casos de uso regulatorios
Comprar 79881-89-3
Comprar 79881-89-3
Encomendar 79881-89-3
Consultar 79881-89-3
price of 79881-89-3
79881-89-3 Fornecedor
79881-89-3 Fabricante
79881-89-3 Exportador
buy high quality Celiprolol Hydrochloride EP Impurity F
Comprar Celiprolol Hydrochloride EP Impurity F
Celiprolol Hydrochloride EP Impurity F fornecedores
Celiprolol Hydrochloride EP Impurity F fabricantes
Celiprolol Hydrochloride EP Impurity F preço
Encomendar Celiprolol Hydrochloride EP Impurity F
Consultar Celiprolol Hydrochloride EP Impurity F
Celiprolol Hydrochloride EP Impurity F custo
Celiprolol Hydrochloride EP Impurity F Fornecedor
Celiprolol Hydrochloride EP Impurity F Distribuidor
Celiprolol Hydrochloride EP Impurity F Para validación de métodos
Celiprolol Hydrochloride Estándar de referencia
Celiprolol Hydrochloride EP Impurity F Para presentación ANDA
Celiprolol Hydrochloride EP Impurity F Para estudios de degradación forzada
Celiprolol Hydrochloride EP Impurity F Estándares de identificación
Celiprolol Hydrochloride EP Impurity F Para presentación DMF
Produtos Relacionados
Isenção de responsabilidade
As informações do produto SynZeal fornecidas neste site são baseadas no entendimento existente no momento da publicação. O cliente é responsável por avaliar a precisão das informações no momento da compra.
A SynZeal atualizará estes detalhes conforme novos desenvolvimentos ou descobertas nas especificações do produto, sem aviso prévio.




